Ozone Removes Iron and Manganese
Iron and manganese removal is one of the more common uses for ozone in drinking water systems. Iron and manganese are easily oxidized by ozone. This document will serve to help understand the fundamentals of iron and manganese oxidation with ozone. We will also cover the practical application of ozone in this application while offering helpful tips learned over the years.
Ozone oxidation of iron and manganese is an extremely fast reaction. In many ozone applications elevated levels of iron and manganese can cause nuisance issues due to soluble iron and manganese inadvertently oxidizing by ozone and dropping out of solution in less than ideal locations. If those concerns are what brought you here, keep reading, we will offer helpful tips to mitigate these issues as best as possible.
Chemistry
Iron and manganese in water cause no health related issues, the main purpose for iron and manganese removal is aesthetics due to the discoloration of water. Removal also may be necessary due to the build-up of iron and manganese on pipes, fixtures and other surfaces.
Both Iron Fe(II) and Manganese Mn(II) are soluble (non-removable) in water causing them to flow directly through conventional filtrations without some form of oxidation to transform them into particulates (removable).
Iron Removal
Soluble Iron Fe(II) is called ferrous iron. Ferrous Iron Fe(II) is oxidized to Ferric Iron Fe(III) by ozone. This Ferric Iron Fe(III) will then hydrolyze to form Fe(OH)3 which is a particulate and can be removed by standard filtration. The reaction of Ferrous Iron Fe(II) to Ferric Iron Fe(II) consumes 0.43 mg of ozone per mg of Fe(II). Iron can also be oxidized by oxygen. Due to the oxidation of iron by oxygen, an ozone system for iron removal may be more efficient than the calculated ozone demand of 0.43 mg ozone per mg iron. The oxidation of ferrous iron requires only an electron exchange and therefore is a fast reaction. The speed of this reaction will typically consume almost all ozone in iron oxidation reaction prior to any manganese oxidation.
Manganese Removal

Soluble Manganese Mn(II) is oxidized by ozone to form manganese dioxide MnO2 which is a particulate and can be easily removed by standard filtration. This process consumes 0.88 mg of ozone per mg of Manganese Mn(II). However, over oxidation of manganese will form soluble permanganate MnO4. While permanganate will normally return to manganese dioxide MnO2 over time (20-30 minutes), it is best to design a manganese removal system with the proper ozone dosages and integrated controls to prevent over oxidation.
Filtration
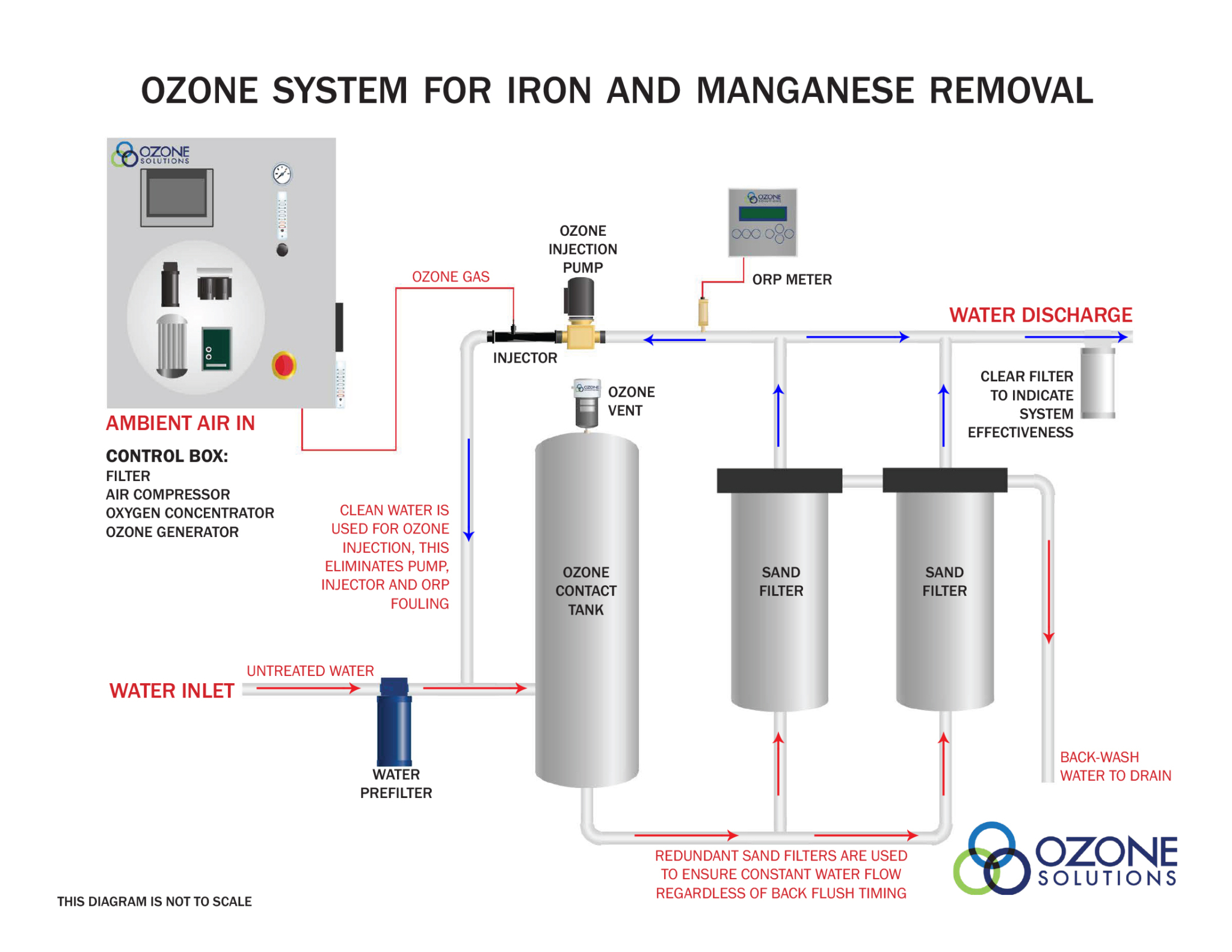
Ozone will oxidize iron and manganese to form insoluble particulates that can easily be filtered from the water. Iron and manganese will build up on the filter over time and must be removed from the process water. A back-washable filter is highly recommended for these applications. Sand filters are widely used for iron and manganese removal due to the simple design and the long lasting filter media. In continuous use systems it will be necessary to use two (2) filters in parallel and time the back-wash cycles to occur at opposite times.
The back-wash water from these filters will have extremely high levels of iron and manganese and must be disposed of with care. While neither iron or manganese have any health or safety risks, there are plumbing considerations to keep in mind as drain pipes may become obstructed with iron and manganese build-up over time.
Practical Application
The use of ozone for iron and manganese removal is very common and has been in use for many years. The reaction of ozone and these metals is fairly simple and straight forward. There are a few design considerations that should be accounted for prior to installing an Ozone System for iron and manganese removal.
System Plumbing
Due to the fast reaction of ozone and iron oxidation there are some important design considerations that must be evaluated prior to system implementation. For an example of a working system design review the diagram below.